Temperature control of photoredox experiments is not easily accomplished. Most experiments are described using a fan to maintain the reaction near room temperature and no fan to let the reaction warm up. Moreover there are very few setups described allowing reaction to be performed below room temperature. In this context it is very challenging to study temperature effect on photocatalysis in a convenient way.
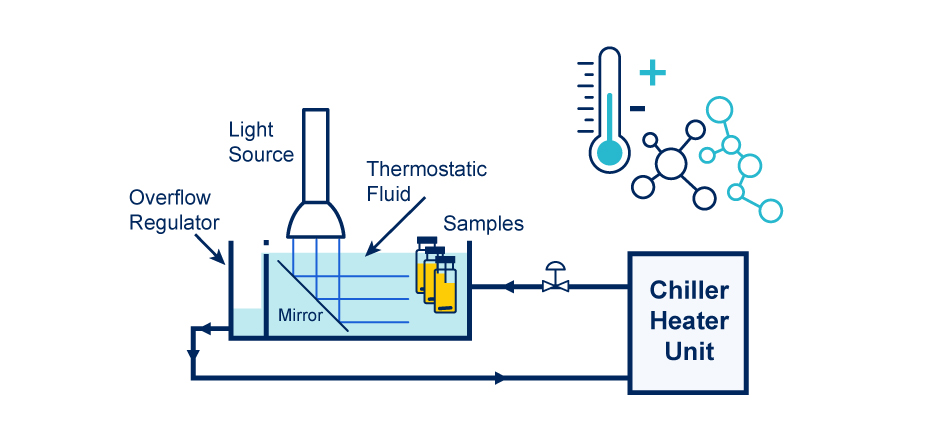
 HepatoChem developed a device PhotoRedOx TC that makes this type of experiments easy to perform using a simple chiller/heater recirculator.
HepatoChem developed a device PhotoRedOx TC that makes this type of experiments easy to perform using a simple chiller/heater recirculator.
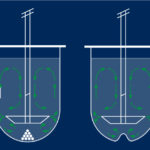
The PhotoRedOx TC (for Temperature controlled) fits many light sources like for example the LEDs EvoluChem 18W. It also has a photochemistry chamber to evenly distribute light. Regarding the format vials, this apparatus is very flexible (from 0.3ml to 20ml) thanks to the availibility of many racks. Flow reactor can also be used with this device. PhotoRedOx TC required to use a magnetic stirring plate to provide agitation and an external fluid circulator to heat or cool the reaction vessel. (light sources, racks, flow reactor, stirring plate and fluid circulator are supplied independently)
In the following examples, HepatoChem shows how CH alkylation using a BF3K reagent can benefit from lowering temperature of the reaction and how increasing the temperature can improve conversion of a C-O cross-coupling.
PhotoRedOx TC (Temperature Controlled)
- Fits many light sources (EvoluChem 18W)
- Photochemistry chamber to evenly distribute light
- Flexible format vials (from 0.3mL to 20mL)
- Flow reactor available
- Stirring on magnetic stirring plate
- External recirculatorneeded to heat or chill reaction vessel
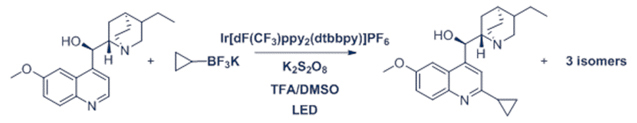
BF3K Cyclopropyl and hydroquinine reaction
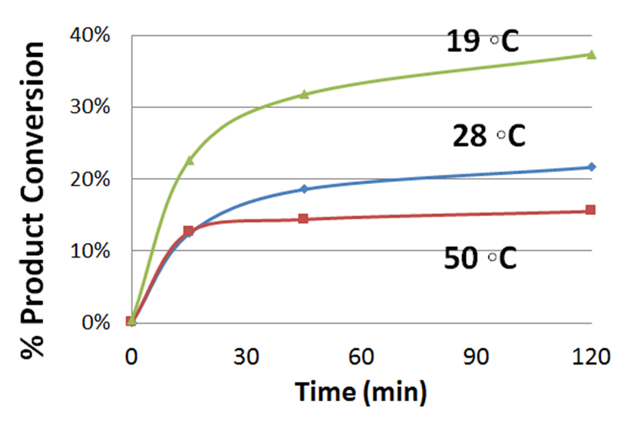
Reaction Conversion at 19°C, 28°C and 50°C
Experimental Details: Reaction performed in Evoluchem Photoredox Temperature control bath with circulating polyethylene glycol/water bath and an EvoluChem 18W 6200K white light for 2 hr. Reaction contains 50 µmol substrate, 1.5 equiv. RBF3K, 2 equiv. K2S2O8, 5 equiv. TFA and 2 mol% Ir(dF-CF3-ppy)2(dtbpy) in 0.5 ml DMSO.
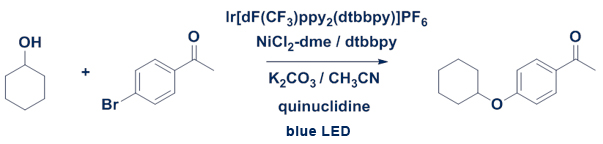
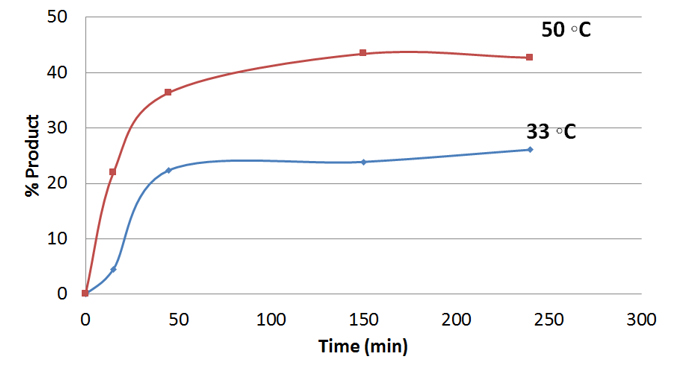
C-O Coupling with Cyclohexanol
Reaction Conversion at 33°C and 50°C
Experimental Details: Reaction performed in Evoluchem Photoredox TC box with EC 450 nm LED. Reaction 2 mol% Ir(dF-CF3-ppy)2(dtbpy), 5 mol% NiCl2-dme/dtbpy and 3 equiv. base with 10 mol% quinuclidine. Conversion determined by LC-UV.
Learn more:
- Find and order the PhotoRedOx TC (temperature controlled) directly on our website
- Find all our products dedicated to PhotoRedOx directly on our website
- Check out our previous posts:
Bring Your Chemistry to Light with the PhotoRedOx Box
Iridium and Nickel Photoredox kits from HepatoChem
Lead Diversification Tool Box from Hepatochem - Contact us: interfine@interchim.fr