The various analytical techniques available today make it possible to obtain accurate and rapid results, but they largely depend on the sample preparation technique.
The aim of sample preparation is to treat the sample matrix and extract the compounds of interest to be analysed.
Today, different techniques are used to process these matrices, which are encountered in various fields such as environmental, toxicology, agri-food and pharmaceutical industries.
Filtration is still the most widely used technique, with syringe filters available in a range of porosities. (see our UptiDisc™ Interchim® range).
In addition, the use of SPE columns (Solid Phase Extraction – PolyClean™ & UptiClean™ Interchim® range), QuEChERS (QuEChERS Interchim® range) and 48 & 96 well plates is common for processing samples with a more complex matrix.
Sample dissolution is also one of those sample preparation techniques mainly found in the pharmaceutical industry.
By definition, sample dissolution is a physico-chemical process by which a solute incorporated into a solvent forms a homogeneous mixture known as a solution.
This technique is essential in the process of manufacturing and qualifying a drug, as it will define the pharmaceutical performance and bioavailability of the active ingredients, which are found in different forms such as tablets, capsules and injectable liquid suspensions.
Setting up a dissolution test requires an understanding of these mechanisms, and remains a non-standardised development technique for all products, as it can change over the course of a drug’s formulation.
Pharmacopoeia regulations define worldwide standardisation criteria for measuring drug release rates in the body.
Overall, dissolution tests will make it possible to:
- optimise therapeutic efficacy during product development and stability assessment
- assess product quality and production to ensure batch-to-batch consistency
- assess biological availability from separate batches of products from one or more manufacturers
- predict in vivo availability (bioavailability)
The automation of dissolution tests on systems such as Agilent, Distek, Erweka, Hanson, Pharmatest and Sotax makes it possible to optimise these methods.
As with any automated system, accessories and consumables are required to ensure that the system operates perfectly.
Interchim® offers a complete range of dissolution equipment compatible with all brands of dissolution equipment used in laboratories (Caleva, Distek, Erweka, Hanson, Pharmatest, Sotax, Vankel, Agilent, etc.).
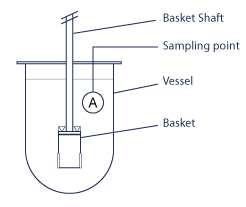
1. The dissolving basket
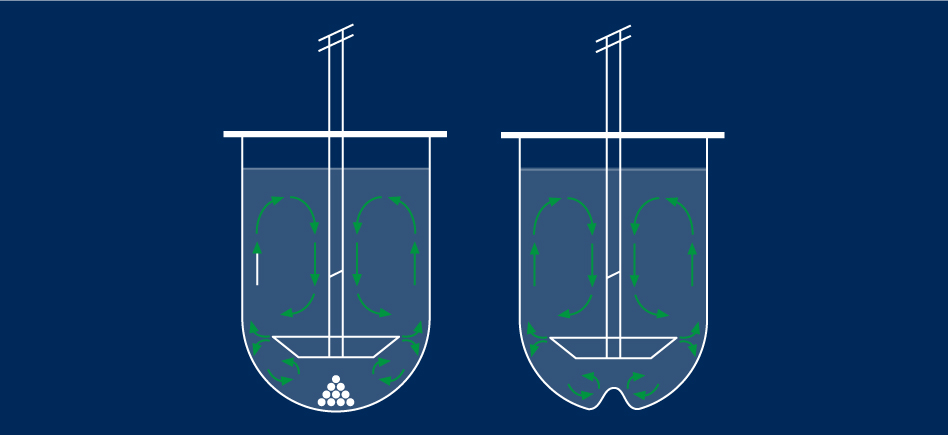
Introduced in 1970, the rotating basket method was the first official method. The apparatus consists of a metal drive shaft connected to a cylindrical basket. The basket is placed inside a container made of glass or another inert, transparent material. The temperature is kept constant by a water bath or heating jacket. The solution in the container is gently agitated by the rotating element.
The USP 1 method requires a 40 mesh screen unless otherwise specified in the monograph. Other mesh sizes can be used to solve individual problems such as clogging by particles or excipients and random product release.
These baskets are available in different shapes and materials.
2. Basket Shaft
This accessory holds the dissolving basket in place while the dissolving bowl is being stirred.
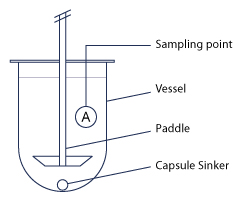
3. Paddles
The paddle is an alternative accessory to the dissolving baskets. The product is placed directly into the dissolving bowl. It is dissolved by the uniform rotary movements of the paddle.
Coatings are available in several materials:
- 316SS stainless steel (polished to eliminate impurities and the risk of corrosion)
- 316SS stainless steel with PTFE coating
- Fluoropolymer
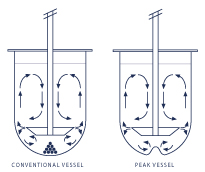
4. Vessels for dissolution
The dissolving bowls are essential for obtaining reproducible and effective results.
Numerous USPs define the dimensions and colours of the dissolving bowls to be used.
Additional accessories include Vessel Covers, Sampling Cannulas and Cannula Filters.
EVER-CLEAN cleaning salts from QLA effectively clean water baths in dissolving systems and prevent the formation of micro-organisms. One dose will keep your bath clean for 8 months.
Learn more:
- Consult our dissolution consumables and accessories on our website
- Download the QLA catalog (Interchim® distribution brand), accessories and consumables for sample dissolution.
- Download the Agilent catalog (Interchim® distribution brand), accessories and consumables for sample dissolution.
- Please do not hesitate to contact us
- Follow our news on LinkedIn