1. Introduction
It is estimated that 20 percent of all pharmaceutical small molecule drugs are halogenated.1 Carbon–halogen bonds lead to a wide range of effects, including an increase in thermal and oxidative stability and an improved biological membrane permeability [1]. Furthermore in drug discovery, natural products (NPs) represent biologically valuable scaffolds and their halogenation may this further improved their properties. A Previous study has demonstrated the potential of chemical halogenation on plant extracts to obtain bioactive compounds [2]. In this context, a strategy for the generic halogenation of plant extracts has been developed to build up libraries of original halogenated NPs in particular for the search of new antifungal agents. To start with, the halogenation reaction was performed with a series of NP standards from different chemical classes. Once the reaction was successfully achieved different plant extracts were submitted to the same generic halogenation procedure (Br, I) [3].
2. Oxidative halogenation of natural products
In this work the idea was to use an eco-friendly halogenation reaction [3] that uses sodium halides in aqueous hydrogen peroxide solution and apply it to a series of pure NPs and crude extracts to estimate if this could increase their antifungal activities.
Halogenation Procedure
- Chloration: 6 eq NaCl – 1g standard – 6eq H2O2 – 10mL CH3COOH – to 25mL with H2O
- Bromation: 6 eq NaBr – 1g standard – 6eq H2O2 – 10mL CH3COOH – to 25mL with H2O
- Iodation: 3 eq NaI – 1g standard – 6eq H2O2 – to 25mL with H2O
- Reaction Time: 24h
- Room temperature
- Under magnetic stirring
- Green chemistry
Chemical reaction
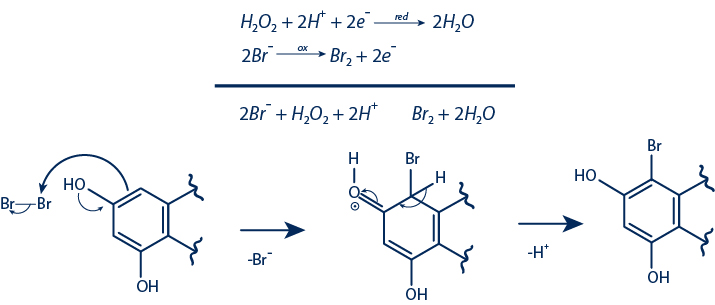
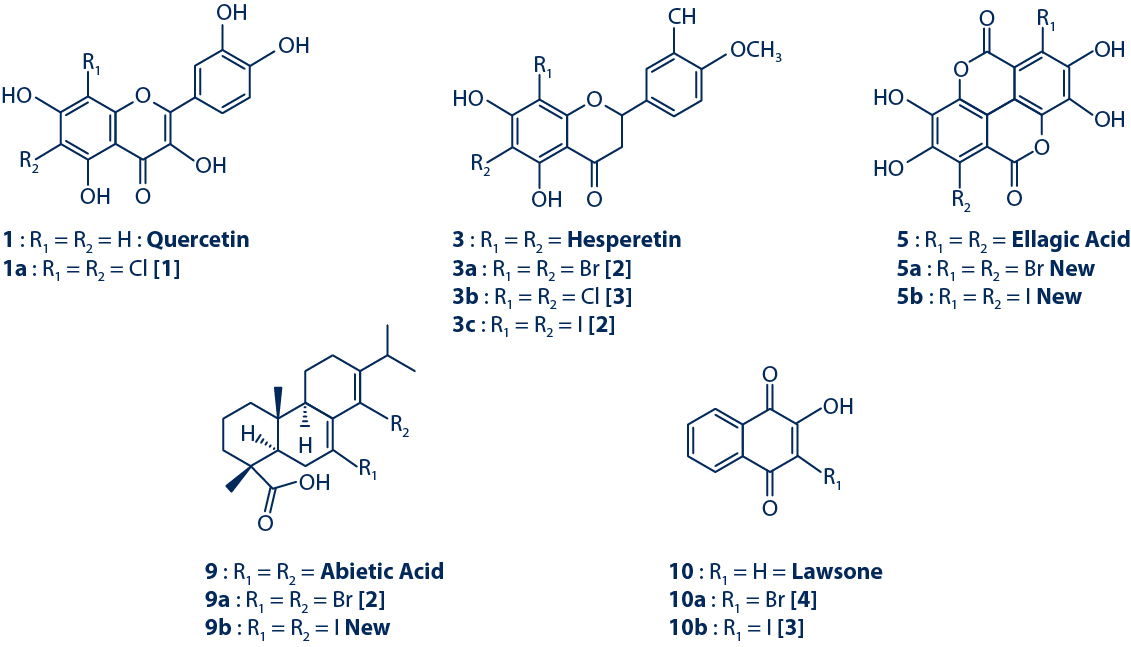
Semisynthetic Compounds Obtained from Oxidative Halogenation
Yield from 0.7% to 29%
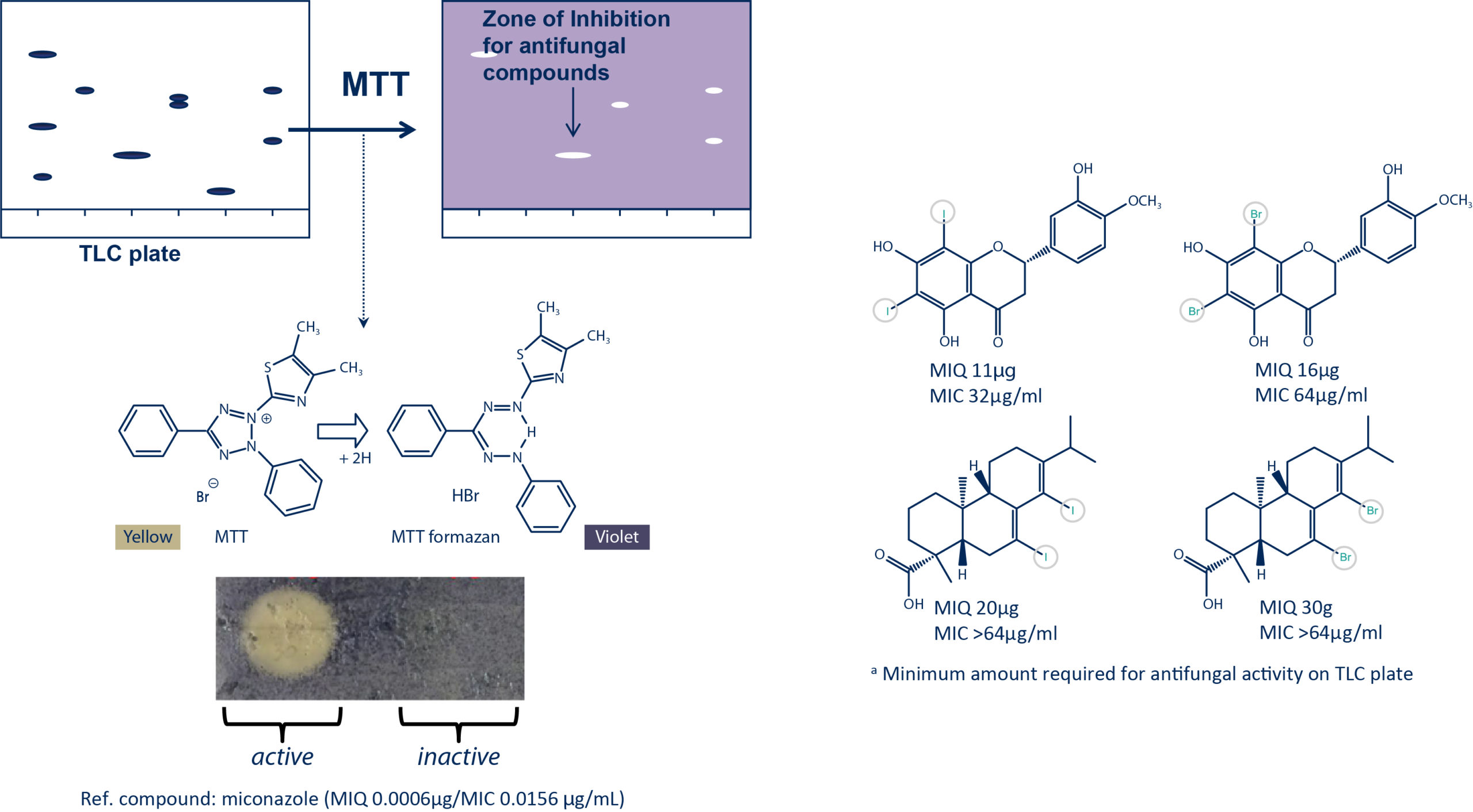
A series of NP standards with different scaffolds were selected for halogenation. The reactions were performed with 50 mg of each compound. Purifications of the halogenated derivatives were achieved by reverse phase semi-preparative HPLC-UV.
3. Antifungal assay against C. albicans
The antifungal activity of the purified halogenated derivatives was evaluated against C. albicans using both the bioautography assay (MIQa) and dilution assay (MIC).
C. albicans
Strains:
CAF 21
DSY 2621
Antifungal bioautography assay
4. Selection of crude extract rich in flavonoids
Citrus species 8.5% world fruits production
 40-60% of oranges are used for fruit juices 50% pericarp on total weight |
Once the halogenation reaction was validated with the series of standards the methodology was applied to a model crude plant extract (Citrus sinensis pericarp) rich in flavonoids and easily obtainable at large scale. |
Citrus sinensis Pericarp consitutents
| Components | wt% on the basis of dry weight |  |
| Ash (%) | 2.56 | |
| Sugar (%) | 9.57 | |
| Fat (%) | 4.00 | |
| Protein (%) | 9.06 | |
| Flavonoid (%) | 4.50 | |
| Pectin (%) | 23.02 | |
| Lignin (%) | 7.52 | |
| Cellulose (%) | 37.08 | |
| Hemicellulose (%) | 11.04 |
5. Bromination of the Citrus pericarp methanolic extract
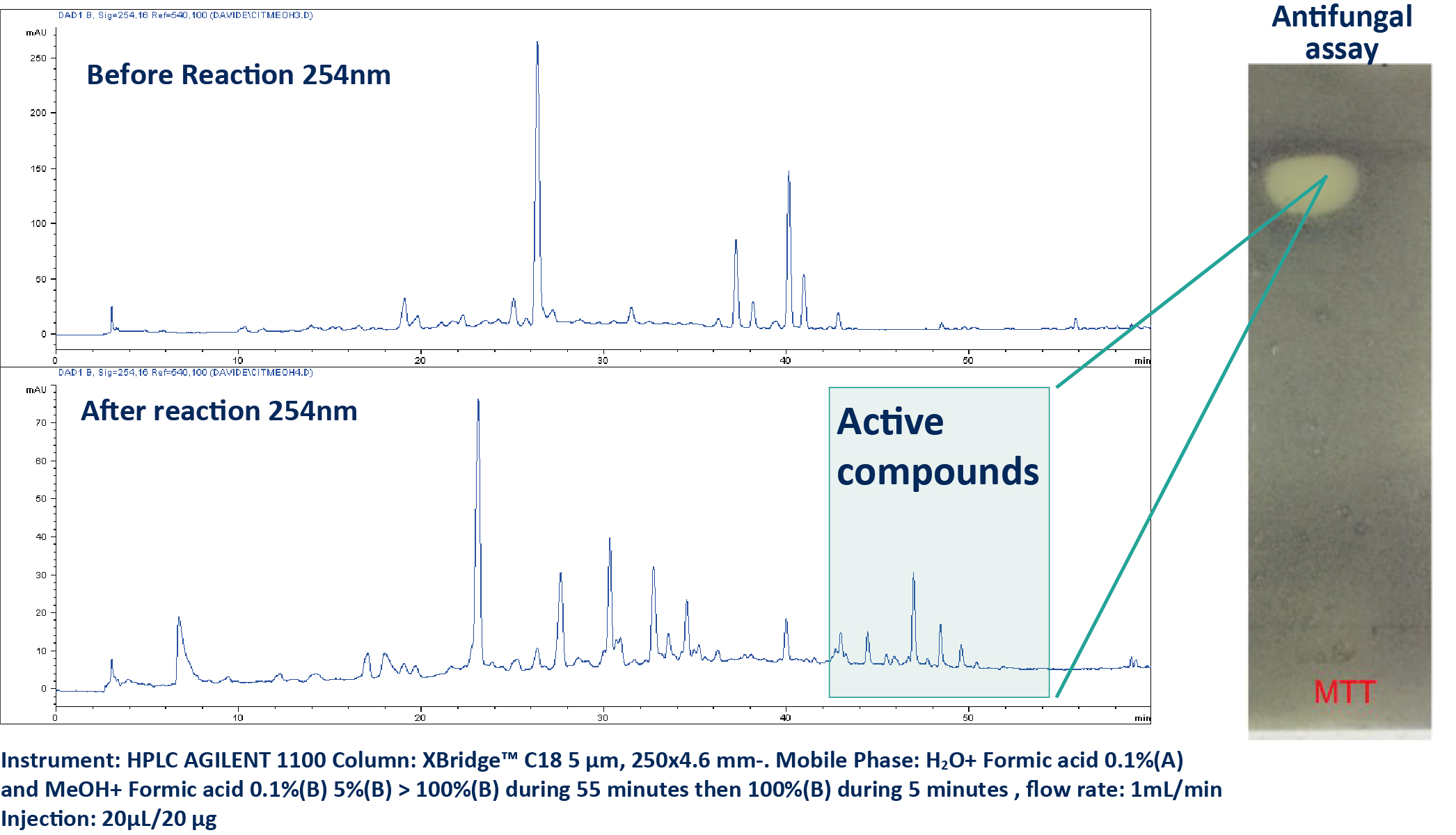
The halogenation of the methanolic extract was first performed at the analytical scale with 100 mg of the extract during 24 hs. After this, the reaction was stopped and controlled by HPLC-UV (A). The crude reaction mixture presented a strong antifungal activity against C. albicans.
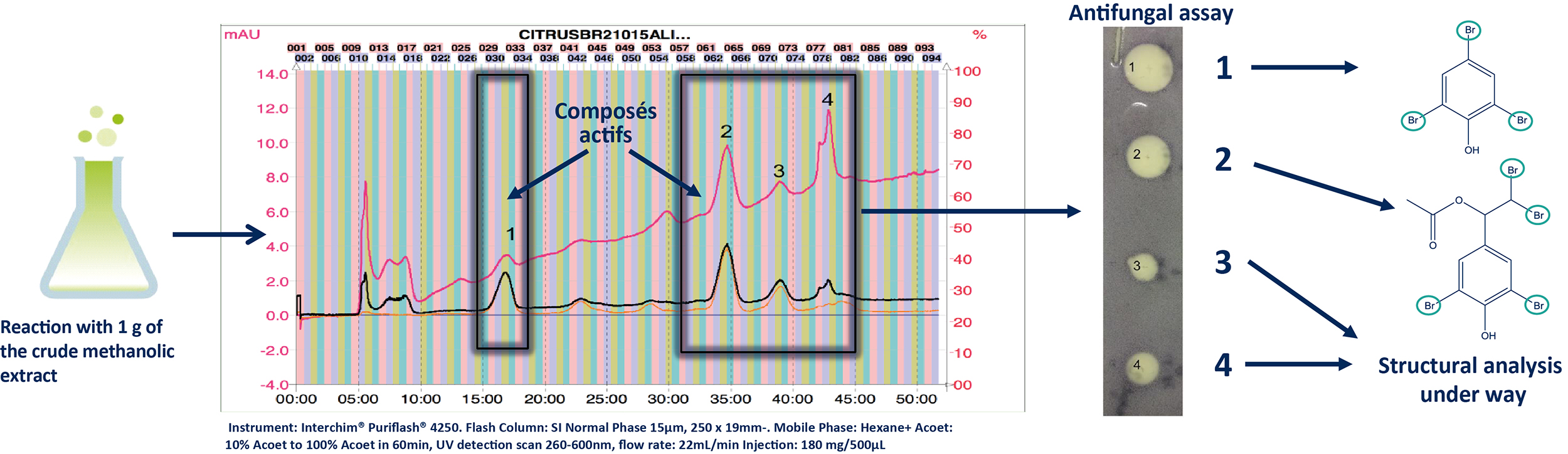
In order to isolate these active compounds the reaction was scaled up to 1 g keeping the same reaction time (24 h) (B). The reaction mixture was dried and purified by normal phase flash chromatography (FC). The preparative conditions were firstly determined at the analytical scale and after then transposed to the preparative scale using the same stationary phase [5]. The fractions obtained were dried and submitted to the antifungal assay allowing to the localization of the four active compounds (1-4). The structural elucidation of these compounds was performed by HRMS and NMR.
a. HPLC UV analysis of the crude extract after and before reaction
b. Normal Phase Ultra Pressure Flash Chromatography (FC-UV)
6. Conclusion
The halogenation of a series of NP standards was found efficient to generate antifungal compounds against the human fungal pathogen Candida albicans. The reaction was conducted with eco-friendly condition using soft reagents. The methodology was also successfully applied to the halogenation of an complex crude plant extract obtained from a waste material (citrus pericarp) generating unusual antifungal halogenated derivatives.
These preliminary results further demonstrate the potential of chemical halogenation of plant extracts to obtain bioactive ‘unnatural’ natural products of interest. In this context the application of such reaction to crude extracts from waste biomass might have a great interest to produce valuable bioactive NPs.
The next step will be the evaluation of the halogenated derivatives against different biological targets and also the application of this methodology to a large number of plant extracts with chemodiverse compositions.
7. References
1. Herrera-Rodriguez et al. Chem Today 2011;29:4
2. Mendez et al. ACS Comb Sci. 2011;13:200-204
3. Bernini et al. New J Chemi. 2015;39:2980-2987
4. Favre-Godal et al. Phytochemistry 2014;105:68-78
5. Challal et al. Planta Med. 2015; 81: 1636-1643
Know more:
 |
|